Clinical Objectives
- To identify normative data of objective motor measures of those body movements that are usually investigated to establish the presence and severity of Parkinson’s disease.
- To identify patients in the preclinical phase of the disease, through a four-step approach:
i) identification of IH through a low-cost standard olfactory test
ii) Measurement of an altered motor pattern in Ideopathic Hypostmia subject, compared to the normal data assessed in an Healthy Control group;
iii) Improvement of motor performance due to dopaminergic stimuli with the pharmacological test with L-dopa; iv) Demonstration of an alteration of the nigro-striatal dopaminergic system using the diagnostic nuclear medicine method SPECT DaTSCAN).
- To use OLIMPIA wearable sensors for the objective analysis of the motor pattern in a clinical environment to assess Parkinson’s Disease patients, in addition to the usual clinical procedure.
- To use OLIMPIA wearable sensors for the objective analysis of the motor pattern at home through the telemedicine service. The instrumentation will also be adapted for use by the patient and / or caregiver. The data of the objective motor pattern will be uploaded on a Cloud database where they will be analysed and the neurologist will verify the correspondence between these data and what is detected by the usual clinical practice.
Clinical Endpoints
Primary Endpoints
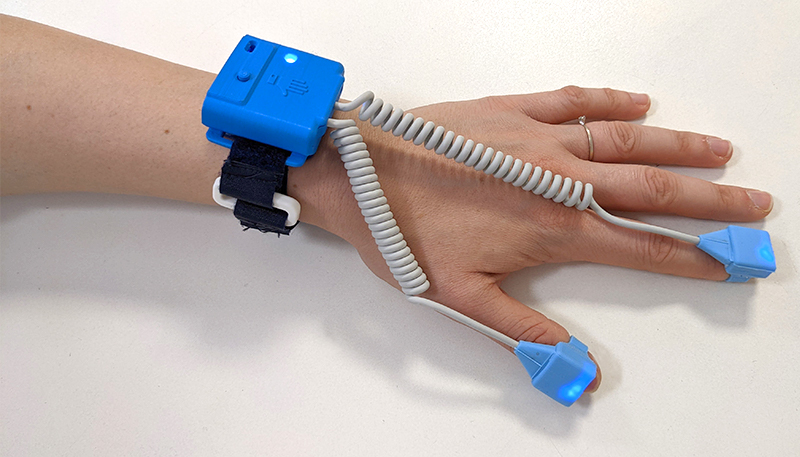
- To acquire motor pattern through the use of OLIMPIA wearable sensors for motion analysis in HC, Ideopathic Hypostmia subjects, Parkinson’s Disease patients, and patients with extrapiramidal syndrome “De-Novo”.
- To obtain normative data of the motor parameters that are clinically relevant. The motion features that are statistically significant in distinguishing between HC and Parkinson’s Disease patients will be identified and their repeatability will be evaluated.
- To correlate normative data with measures obtained by Ideopathic Hypostmia subjects. The demonstration of a subtle deficit in motor performance that is not clinically evident could anticipate the disease cardinal symptoms by some years. The preclinical diagnosis will be investigated through the biomechanical analysis provided by the OLIMPIA wearable sensors in IH subjects, which are at risk for developing Parkinson’s Disease.
- To correlate the clinical total data with parameters obtained by the nuclear medicine diagnostic (i.e., SPECT DaTSCAN).

Secondary Endpoints
- Clinical control (in hospital) through objective motor measures.
- Clinical control (at home) via telemedicine with wearable wireless instrumentation. Correlate the motor measures measured at the hospital with those detected by patients at home.
Recruitment
- 150 Healthy Subjects , [75 female, 75 male], as control group, enrolled by the General Practitioners, as normosmic at the standard olfactory test.
- 100 Ideopathic Hypostmia subjects, recruited by the General Practitioners through the administration of the standard olfactory test.
- 50 Parkinson’s Disease patients [25 F, 25 M], of which 15 PD patients will be assessed with the wearable system also at home during the final months of the experimentation.
- 30 De-Novo Parkinson’s Disease patients drug-naïve, who will be subjected to the sub-acute challenge dopaminergic test with L-dopa.